数字化转型网数据专题将关注数据治理、数据质量管理、数据架构、主数据管理、数据仓库、元数据管理、数据备份、数据挖掘、数据分析、数据安全、大数据、数据合规、等数据相关全产业链相关环节。

原文于 November 7, 2024 发表在PharmaTech,作者:Chris Burgess
Nomenclature is important. Data governance, data integrity, and data quality are all widely used terms, but what do they actually mean and how are they connected? The purpose of this article is to provide a structured model for these terms with their definitions and their relationships in the context of analysis and testing within a pharmaceutical quality system.
术语很重要。数据治理、数据完整性和数据质量都是广泛使用的术语,但它们实际上是什么意思,它们之间又是如何关联的呢?本文的目的是提供一个结构化的模型,用于这些术语的定义以及它们在药品质量系统分析测试背景下的关系。
The crucial concept is that data quality, including data integrity, is only attainable via data governance, as will be illustrated in the proposed model.
关键概念是,数据质量,包括数据完整性,只有通过数据治理才能实现,正如所提出的模型将展示的那样。
Introduction
In a regulatory context, the establishment of measured values and subsequent reportable results of a predefined quality is an essential activity. Reportable results are then compared with predetermined acceptance criteria and/or standards and specifications. The processes, mechanisms, and control systems necessary to establish measurement values and reportable results of a defined quality are interlinked. These interlinkages may include metrological, procedural, or organizational elements
在监管环境中,建立测量值和随后的预定义质量报告结果是一项基本活动。报告结果随后与预定的验收标准、规范和/或技术规格进行比较。建立测量值和预定义质量报告结果所需的过程、机制和控制系统是相互关联的。这些相互关联可能包括计量学、程序或组织要素。
The overall proposed model is built up using building blocks akin to the construction of Lego brick models. This approach has been used in previous papers concerning error budgets in measurement uncertainty and Monte Carlo Simulation, and data quality within a lifecycle approach
(1–2).整体提出的模型是使用类似于乐高积木模型构建的模块构建而成的。这种方法已在关于测量不确定性的误差预算和蒙特卡洛模拟以及生命周期方法中的数据质量方面的先前论文中使用过(1-2)。
An analytical testing flow diagram, shown in Figure 1, gives a high-level idea of the traceability and subsequent interactions from the marketing authorization (new drug application, NDA, or abbreviated new drug application, ANDA) to the reportable result and data quality.
一个分析测试流程图,如图 1 所示,提供了从市场许可(新药申请,NDA 或简化新药申请,ANDA)到报告结果和数据质量的可追溯性和后续交互的高级概念。
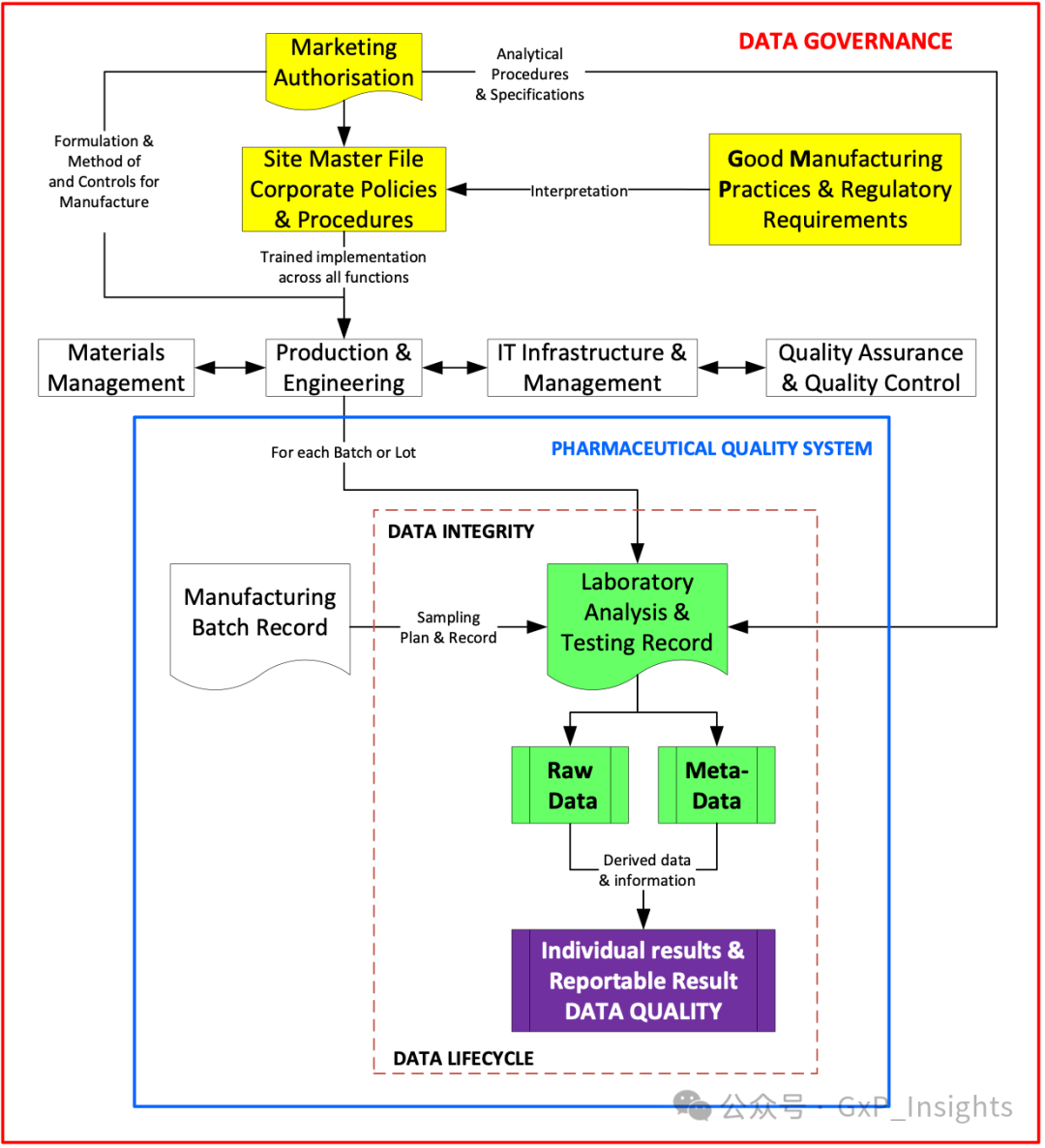
Figure 1: A high-level process flow indicating some of the elements of data governance, data quality, and data integrity, as well as the quality management system (QMS).
图 1:表示数据治理、数据质量和数据完整性的一些要素以及质量管理体系(QMS)的高级流程图。
This process flow has been translated into the Lego brick model shown in Figure 2. This model is based upon a data quality within a lifecycle approach model which has been modified and extended, as will be described in some detail (2).
此流程图已转换成图 2 所示的乐高积木模型。此模型基于生命周期方法模型中的数据质量,经过修改和扩展,将在某些细节中详细描述(2)。
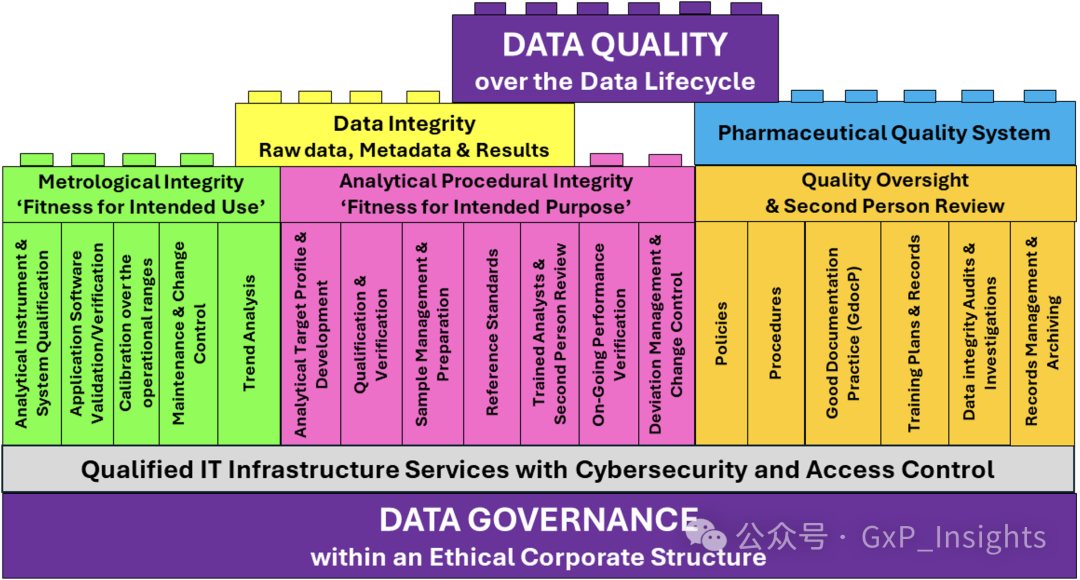
Figure 2: Data governance model modified and extended from the data quality within a lifecycle approach model (taken from reference 2, Figure 15).
图 2:从生命周期方法模型中的数据质量改进和扩展的数据治理模型(参考 2,图 15)。
The crucial concept is that data quality is only attainable via data governance.
关键概念是,数据质量只能通过数据治理来实现。
The data governance and data quality Lego brick model
数据治理与数据质量乐高积木模型
Data quality is not an accident but a product of design. It is a combination of data integrity and the functionality and control under the Pharmaceutical Quality System, usually termed the quality management system (QMS). These aspects are underpinned by data governance and qualified information technology (IT) infrastructure services. This is essentially a sandwich structure in which the “filling” is provided by metrological integrity, analytical procedure integrity, and quality oversight. These “fillings” are described below.
数据质量并非偶然,而是设计的结果。它是数据完整性、药品质量体系下的功能性和控制力的结合,通常被称为质量管理体系(QMS)。这些方面由数据治理和合格的信息技术(IT)基础设施服务支撑。这本质上是一种三明治结构,其中“填充物”由计量完整性、分析程序完整性和质量监督提供。以下是对这些“填充物”的描述。
All the elements shown in Figure 2 are subject to risk assessments and risk management (3), which will not be discussed here
所有图 2 中显示的元素均需进行风险评估和管理(3),此处不予讨论。
The qualified IT infrastructure services with cybersecurity and access control, as seen above, also will not be covered further in this article.
合格的 IT 基础设施服务,如上所述,具有网络安全和访问控制,本文将不再进一步介绍。
It is, however, necessary to discuss the key elements of data integrity and the Pharmaceutical Quality System which, when combined, generate data quality on the foundation of data governance.
然而,讨论数据完整性的关键要素以及药品质量体系是必要的,这两者结合在一起,在数据治理的基础上产生数据质量。
Data governance is the totality of arrangements to ensure that data, irrespective of the format in which they are generated, are recorded, processed, retained, and used to ensure a complete, consistent, and accurate record throughout the data lifecycle
(4).数据治理是指确保数据,无论其生成格式如何,都被记录、处理、保留并用于确保在整个数据生命周期中保持完整、一致和准确的记录的总体安排(4)。
Data integrity
The purpose of an analytical procedure is to provide a reportable result of the analytical characteristic or quality attribute being determined. Analysis and testing require a measurement system, and a procedure for its application to a sample.
分析程序的目的在于提供所确定的特性或质量属性的报告结果。分析和测试需要一套测量系统及其应用于样品的程序。
Data integrity is underpinned by the first brick, the metrological integrity of the instrument or system’s operational performance, with demonstrable assurance that it is “fit for intended use” within a specific analytical procedure, which is “fit for intended purpose” over the data lifecycle.
数据完整性由第一块基石支撑,即仪器或系统的计量完整性及其操作性能,通过可证明的保证,在特定的分析程序中“适用于预期用途”,在整个数据生命周期中“适用于预期目的”。
Metrological integrity
计量完整性
Analysis and testing usually involve the use of an apparatus, analytical instrument, or system to make a measurement. Therefore, establishment of “fitness for intended use” for any apparatus, analytical instruments, or systems used in analysis and testing is necessary to ensure metrological integrity over the operational ranges required.
分析和测试通常涉及使用仪器、分析仪器或系统进行测量。因此,对于在分析和测试中使用的任何仪器、分析仪器或系统建立“适用于预期用途”是必要的,以确保在所需操作范围内保持计量完整性。
Therefore, it is essential to establish “fitness for intended use” before the analytical procedure is performed. The main resource for instrument and system requirements are the specific monographs and general chapters in the pharmacopeias. In particular, United States Pharmacopeia (USP) has a unique general chapter on the lifecycle processes and requirements for ensuring that any apparatus, analytical instrument, or system is “fit for intended use,” as seen in Figure 3 (5).
因此,在执行分析程序之前,建立“适合预期用途”是至关重要的。仪器和系统要求的主要资源是药典中的具体专论和总论。特别是,美国药典(USP)有一个独特的总论,关于生命周期过程和确保任何设备、分析仪器或系统“适合预期用途”的要求,如图 3(5)所示。

Figure 3: Data quality outline for an analysis and testing quality control (QC) model using USP references.
图 3:使用 USP 参考的分析和测试质量控制(QC)模型的数据质量概述。
Assurance lifecycle activities include:
保证生命周期活动包括:
- analytical instrument and system qualification
- 分析仪器和系统确认
- application software validation
- 应用程序软件验证
- calibration over the operational ranges of critical measurement functions
- 校准关键测量功能的操作范围
- maintenance and change control
- 维护和变更控制
- trend analysis to monitor an ongoing state of control.
- 趋势分析以监控持续的控制状态。
The second component of data integrity is a validated or verified analytical procedure performed by a trained analyst.
数据完整性的第二个组成部分是由受过培训的分析员执行的验证或验证过的分析程序。
Analytical procedure integrity
分析程序完整性
It has been a requirement for more than 20 years that analytical methods and procedures need to be validated or verified (6–7). Recently, these requirements have been updated by the International Council for Harmonisation (ICH) and a new guideline on analytical procedure development issued (8–9). USP General Chapter <1220> on Analytical Procedure Lifecycle should be consulted (10).
已超过 20 年,分析方法和程序需要得到验证或确认(6–7)。最近,这些要求已被ICH更新,并发布了关于分析程序开发的新指南(8–9)。应查阅《美国药典》《1220》关于分析程序生命周期(10)。Critical lifecycle activities include:关键生命周期活动包括:
- analytical target profile and development
- 分析目标档案和发展
- qualification and verification
- 确认和验证
- sample management and preparation
- 样本管理和准备
- use of reference standards
- 参考标准的使用
- trained analysts and second person review
- 培训分析师和第二人审核
- ongoing performance verification
- 持续的性能验证
- deviation management and change control.
- 偏差管理与变更控制。
Particular attention should be taken regarding analyst training and second person review within the laboratory.
应特别关注实验室内的分析师培训和第二人审核。
Second person review is essential in ensuring data quality.
第二人审核对于确保数据质量至关重要。
The Pharmaceutical Quality System
药品质量体系
Quality control is the guardian of scientific soundness, whereas the quality assurance function is the guardian of compliance. To perform this duty of care, the quality assurance function requires a robust and comprehensive quality management system that enshrines the elements to provide and perform the necessary quality oversight over the data lifecycle.
质量控制是科学严谨的守护者,而质量保证功能是合规性的守护者。为了履行这一谨慎义务,质量保证功能需要一个强大而全面的质量管理体系,该体系将提供和执行必要质量监督的要素融入其中。
Quality oversight involves both reviewing and auditing activities of QC but also includes the Pharmaceutical Quality System implementation itself to ensure that it is up to date (11).
质量管理监督包括对 QC 的审查和审计活动,同时也包括药品质量体系的实施本身,以确保其保持最新状态(11)。
Quality oversight covers key areas such as:
质量管理涵盖关键领域,例如:
- policies
- 政策
- procedures
- 程序
- good documentation practice (GDocP)
- 良好的文档实践(GDocP)
- training plans and records
- 培训计划和记录
- data integrity audits and investigations
- 数据完整性审计和调查
- records management and archiving
- 记录管理与归档
- second person review (12).
- 第二人审核(12)。
ALCOA models for data integrity
ALCOA 模型用于数据完整性
Much has been written on this topic, particularly regarding the three ALCOA models and the meanings of their acronyms (4). These acronyms are summarized below and illustrated in Figure 4.
关于这个主题已经有很多文献讨论,尤其是关于三个 ALCOA 模型及其缩写的含义(4)。以下是对这些缩写的总结,并在图 4 中进行了说明。
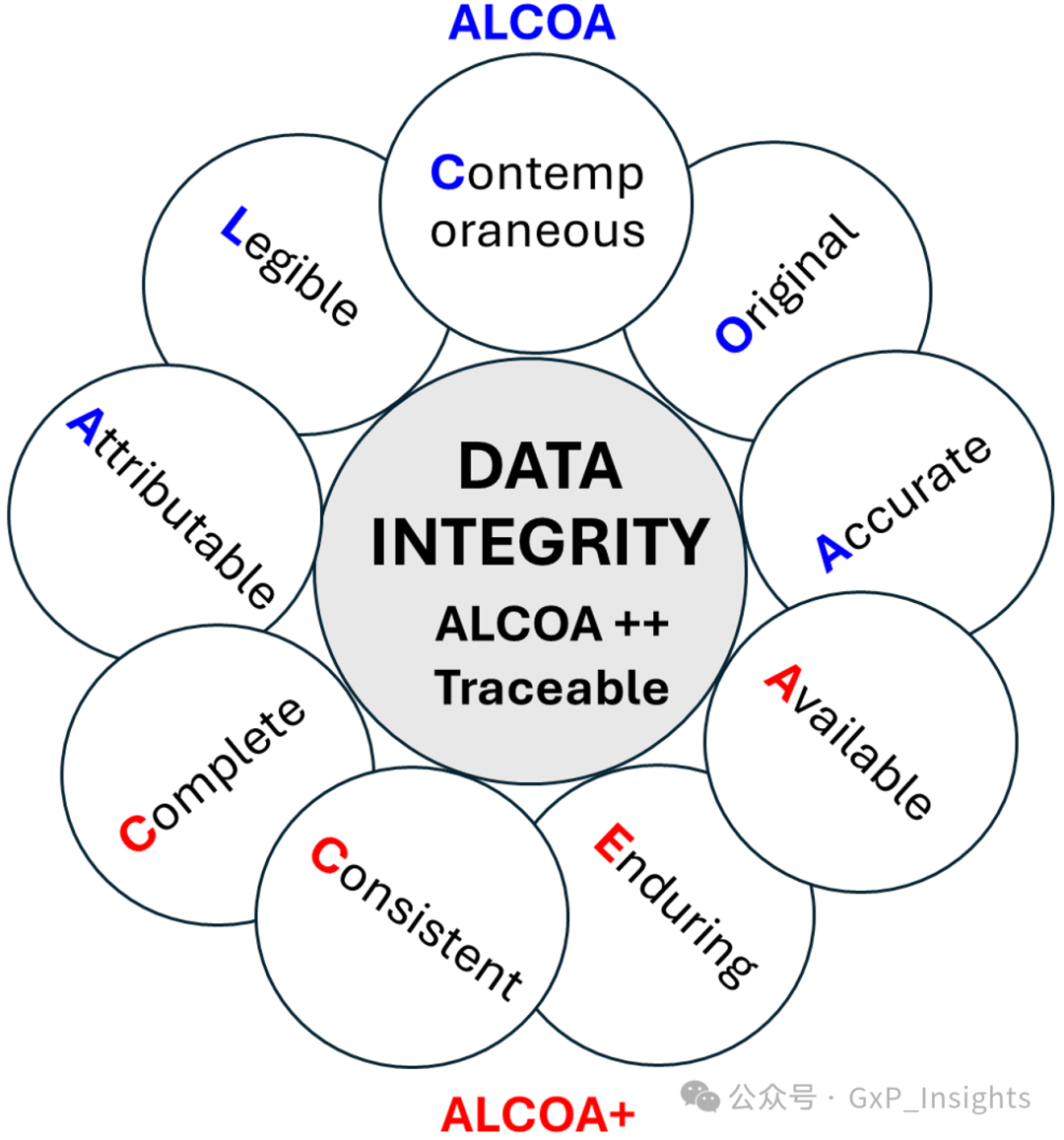
Figure 4: Pictorial representation of the three ALCOA models.
图 4:三种 ALCOA 模型的图示表示。
ALCOA (13)
Attributable It must be possible to identify the individual or computerized system that performed a recorded task, and when the task was performed. This also applies to any changes made to records, such as corrections, deletions, and changes, where it is important to know who made a change, when, and why.
必须能够识别执行记录任务的个人或计算机化系统,以及任务执行的时间。这也适用于对记录所做的任何更改,例如更正、删除和更改,了解谁进行了更改、何时以及为什么很重要。
Legible All data, including any associated metadata, should be unambiguously readable throughout the lifecycle. Legibility also extends to any changes or modification to the original data made by an authorized individual so that the original entry is not obscured.
所有数据,包括任何相关元数据,应在其整个生命周期内清晰可读。可读性也扩展到由授权个人对原始数据进行任何更改或修改的情况,以确保原始条目不被遮挡。
Contemporaneous Data should be recorded on paper or electronically at the time the observation is made. All data entries must be dated and signed by the person entering the data.
数据应在观测时以纸质或电子形式记录。所有数据条目必须注明日期并由录入数据的人员签名。
Original The original record is the first capture of information, whether recorded on paper (static) or electronically (usually dynamic, depending on the complexity of the system). Data or information originally captured in a dynamic state remain in that state.
原始记录是信息的首次捕获,无论记录在纸上(静态)还是电子形式(通常动态,取决于系统的复杂性)。原本以动态状态捕获的数据或信息保持在该状态。
Accurate Records need to be a truthful representation of facts to be accurate. No errors in the original observation(s) and no editing are allowed without documented amendments or audit trail entries by authorized personnel. Accuracy is assured and verified by a documented review including review of audit trails.
记录必须是事实的真实反映,以确保准确性。原始观察中不得有错误,未经授权人员记录的修正或审计跟踪条目不得进行编辑。准确性通过包括审计跟踪审查的文件审查得到保证和验证。
ALCOA+ (14)
Complete All data from an analysis, including any data generated including original data, data before and after repeat testing, reanalysis, modification, recalculation, reintegration, and deletion. For hybrid systems, the paper output must be linked to the underlying electronic records used to produce it. A complete record of data generated electronically includes relevant metadata.
所有分析数据,包括生成的任何数据,包括原始数据、重复测试前后的数据、重新分析、修改、重新计算、重新整合和删除的数据。对于混合系统,输出必须与用于生成它的底层电子记录相关联。电子生成数据的完整记录包括相关元数据。
Consistent Data and information records should be created, processed, and stored in a logical manner that has a defined consistency. This includes policies or procedures that help control or standardize data (such as chronological sequencing, date formats, units of measurement, approaches to rounding, significant digits, etc.).
数据和记录应按逻辑方式创建、处理和存储,并具有定义一致性的逻辑方式。这包括帮助控制或标准化数据(如时间顺序、日期格式、计量单位、舍入方法、有效数字等)的政策或程序。
Enduring Data are recorded in a permanent, maintainable, authorized media form during the retention period. Records should be kept in a manner such that they continue to exist and are accessible for the entire period during which they are needed. They need to remain intact as an indelible and durable record throughout the record retention period.
数据在保留期内以永久、可维护、授权的媒体形式记录。记录应保持其存在并可在整个所需期间内访问。在整个记录保留期内,它们需要保持完整,作为不可磨灭和耐用的记录。
Available Records should be available for review at any time during the required retention period, accessible in a readable format to all applicable personnel who are responsible for their review, whether for routine release decisions, investigations, trending, annual reports, audits, or inspections.
记录应在所需保留期内随时可供审查,以可读格式供所有负责审查的相关人员访问,无论用于常规发布决策、调查、趋势分析、年度报告、审计或检查。
ALCOA++
Traceable Data should be traceable though the lifecycle. Any changes to data or metadata should be explained and should be traceable without obscuring the original information. Timestamps should be traceable to a trusted time source. Metrological standards and instrument or system qualification should be traceable to international standards wherever possible.
数据应在其生命周期内可追溯。对数据或元数据的任何更改都应进行解释,并且应在不掩盖原始信息的情况下可追溯。时间戳应可追溯至可信的时间源。在可能的情况下,计量标准和仪器或系统鉴定应可追溯至国际标准。
Data quality
Data quality is a combination of data integrity and overall control as part of the pharmaceutical quality system.
数据质量是药品质量系统的一部分,是数据完整性和总体控制的组合。An example of a quality control data quality outline for analysis and testing, using examples from USP, is illustrated in Figure 3.
一个用于分析和测试的质量控制数据质量大纲示例,使用 USP 的例子,如图 3 所示。
Summary
摘要
Data quality cannot be assured without a data governance structure supported by a qualified IT infrastructure services with cybersecurity and access control.
数据质量无法得到保证,除非有合格的 IT 基础设施服务支持的数据治理结构,包括网络安全和访问控制。The proposed Lego brick model provides a structural framework for assuring data quality over the lifecycle.
建议的乐高积木模型为在整个生命周期内确保数据质量提供了一个结构框架。
声明:本文来自PharmaTech,版权归作者所有。文章内容仅代表作者独立观点,不代表数字化转型网立场,转载目的在于传递更多信息。如有侵权,请联系我们。数字化转型网www.szhzxw.cn
数字化转型网数据专题包含哪些内容
数字化转型网数据专题将关注数据治理、数据质量管理、数据架构、主数据管理、数据仓库、元数据管理、数据备份、数据挖掘、数据分析、数据安全、大数据、数据合规、等数据相关全产业链相关环节。
数字化转型网数据专题包含: 数字化转型网(www.szhzxw.cn)
1、数据相关外脑支持:100+数据相关专家、100+数据实践者、1000+相关资料
2、数据研习社:与全球数据相关专家、实践者共同探讨相关问题,推动产业发展!
3、国际认证培训:目前已引进DAMA国际认证CDMP,其他国内外认证也在逐步引进中
4、典型案例参考:与数字化转型网数据要素X研习社社员一起学习典型案例,共探企业数据落地应用

本文由数字化转型网(www.szhzxw.cn)转载而成,来源于PharmaTech,作者:Chris Burgess;编辑/翻译:数字化转型网默然。






